Because a multiple bond is counted as a single bond in the VSEPR model, each carbon atom behaves as if it had two electron groups. The next two carbon atoms share a triple bond, and each has an additional single bond. Use Figure 5.1.3 to determine the molecular geometry around each carbon atom and then deduce the structure of the molecule as a whole.īecause the carbon atom on the left is bonded to four other atoms, we know that it is approximately tetrahedral. Use the VSEPR model to predict the molecular geometry of propyne (H 3C–C≡CH), a gas with some anesthetic properties.Ĭount the number of electron groups around each carbon, recognizing that in the VSEPR model, a multiple bond counts as a single group. We can therefore predict the CH 3–N portion of the molecule to be roughly tetrahedral, similar to methane: The four bonds around carbon mean that it must be surrounded by four bonding electron pairs in a configuration similar to AX 4. We can treat methyl isocyanate as linked AX mE n fragments beginning with the carbon atom at the left, which is connected to three H atoms and one N atom by single bonds. In addition, there was significant damage to livestock and crops. The resulting highly exothermic reaction caused a rapid increase in pressure that ruptured the tanks, releasing large amounts of methyl isocyanate that killed approximately 3800 people and wholly or partially disabled about 50,000 others. In 1984, large quantities of Sevin were accidentally released in Bhopal, India, when water leaked into storage tanks. We will demonstrate with methyl isocyanate (CH 3–N=C=O), a volatile and highly toxic molecule that is used to produce the pesticide Sevin. The VSEPR model can be used to predict the structure of somewhat more complex molecules with no single central atom by treating them as linked AX mE n fragments. 
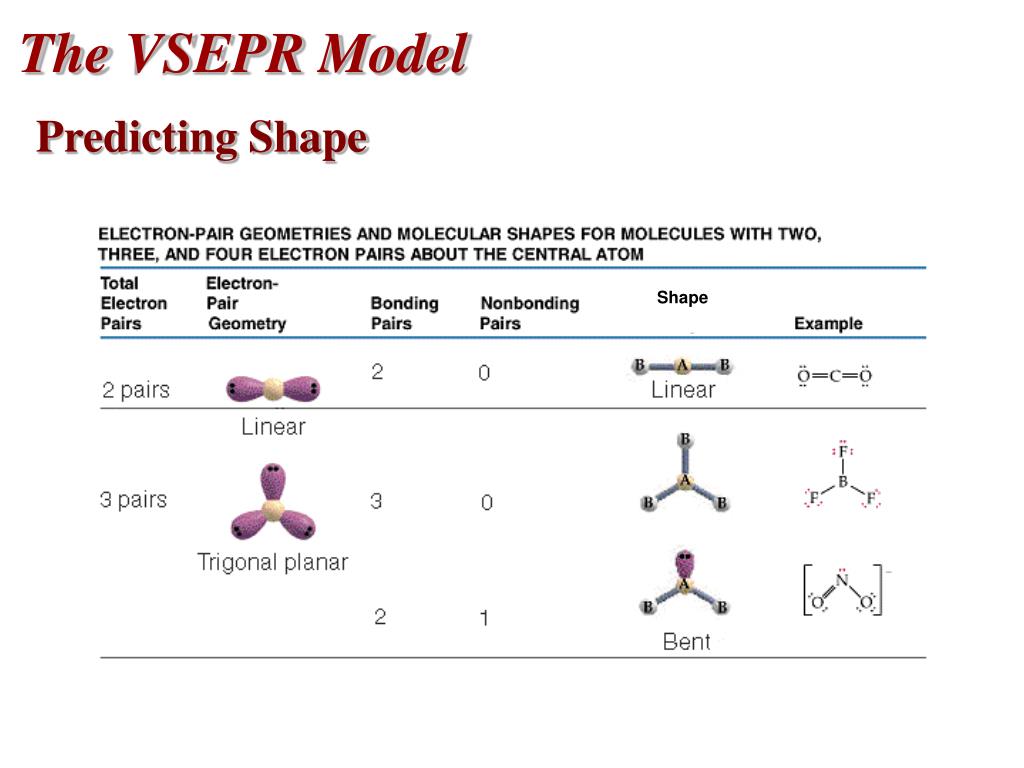
In our discussion we will refer to Figure 5.1.2 and Figure 5.1.3, which summarize the common molecular geometries and idealized bond angles of molecules and ions with two to six electron groups. We will illustrate the use of this procedure with several examples, beginning with atoms with two electron groups. Assign an AX mE n designation then identify the LP–LP, LP–BP, or BP–BP interactions and predict deviations from ideal bond angles.Determine the electron group arrangement around the central atom that minimizes repulsions.Draw the Lewis electron structure of the molecule or polyatomic ion.Using this information, we can describe the molecular geometry The arrangement of the bonded atoms in a molecule or a polyatomic ion in space., the arrangement of the bonded atoms in a molecule or polyatomic ion. From the BP and LP interactions we can predict both the relative positions of the atoms and the angles between the bonds, called the bond angles The angle between bonds. Each group around the central atom is designated as a bonding pair (BP) or lone (nonbonding) pair (LP). In the VSEPR model, the molecule or polyatomic ion is given an AX mE n designation, where A is the central atom, X is a bonded atom, E is a nonbonding valence electron group (usually a lone pair of electrons), and m and n are integers. 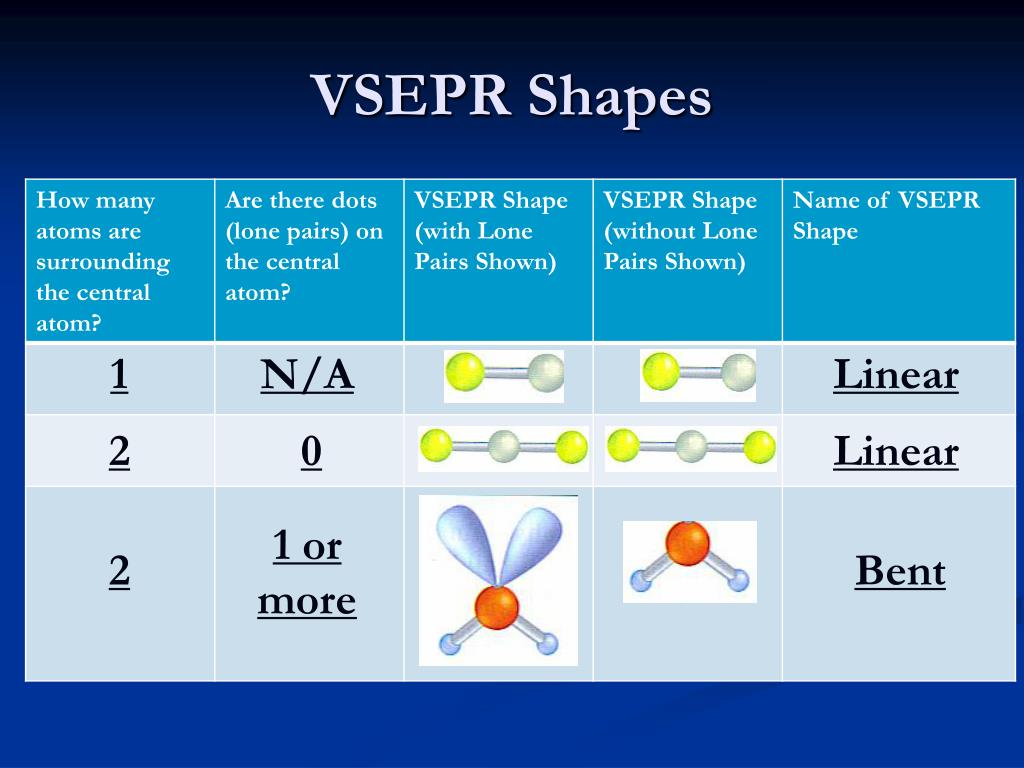
That is, the one that minimizes repulsions. The VSEPR model has gained widespread acceptance as a pedagogical tool and has been further refined to consider the shapes and sizes of electron pair domains, as well as the influence of the core of an atom on molecular geometry.\)įigure 5.1.2 Geometries for Species with Two to Six Electron Groupsįigure 5.1.1 Common Structures for Molecules and Polyatomic Ions That Consist of a Central Atom Bonded to Two or Three Other Atoms Groups are placed around the central atom in a way that produces a molecular structure with the lowest energy. The electron density, which can be calculated using ab initio or density functional theory methods, provides information about bonding and can be obtained experimentally through X-ray crystallography. The VSEPR model can be applied at different levels of sophistication, from a purely empirical set of rules to more advanced treatments based on the analysis of electron density. This leads to specific arrangements of atoms in a molecule, such as linear, trigonal planar, tetrahedral, and so on. 
The model suggests that these electron pairs behave as if they repel each other and try to keep as far apart as possible. The VSEPR model can be used to predict the molecular geometry of a molecule by considering the repulsion between valence shell electron pairs. How can the VSEPR model be used to predict the molecular geometry of a molecule? 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
